- Blog
- Battle tank ascii text art
- Free google sheets expense template monthly
- Stick ranger cavern 2
- Avigilon control center player free
- Battleship game online free against computer
- Sanctuary rpg black edition save editor
- Dragon age inquisition glyphs ancient baths
- Density of water in g
- 2048 x 1152 cool youtube banners
- Florida time zone map
- Ps3 eye camera driver windows 10
- Is decibels a logarithmic scale
- Bohemian rhapsody sheet music piano easy pdf
- Who developed the atomic theory
- What window command copies text
- Cavalier king charles spaniel seattle rescue
- Stanley sharpshooter staple gun not working
- Transparent background crosshairs png
- Space shuttle endeavour simulator ride
- Online color picker from image
- Best automatic pool covers
- Remington model 12 serial number dates
- Red hair highlighting coloring ideas
- Medicare abcd cost
- Google chrome webstore themes
- Space shuttle endeavour vertical display
- Play bejeweled 2 deluxe free
- Play game 2048 online
- Pc anime dancing live wallpaper free download
- Implementation of AGV robots
- Ivory ella elephant ivory ella elephant outline
- Mp3 to midi free converter download
- Free coins for solitaire tripeaks
- Gta v zombie apocalypse mod
- World of warships tanz skins
- Free licensed practical nurse resume template downloads
- Tattoo font letter examples
- Adage script font free
- Professional word document resume template
- Wedding ring box
- Frcp motion to dismiss
- Propresenter background loops free
- Queen victoria hemophilia family tree
- Rfactor 2 dx12 night tracks
- Hot incent sex stories in urdu
- Transfer media files from android to windows
- Free printable coordinate graphing pictures worksheets
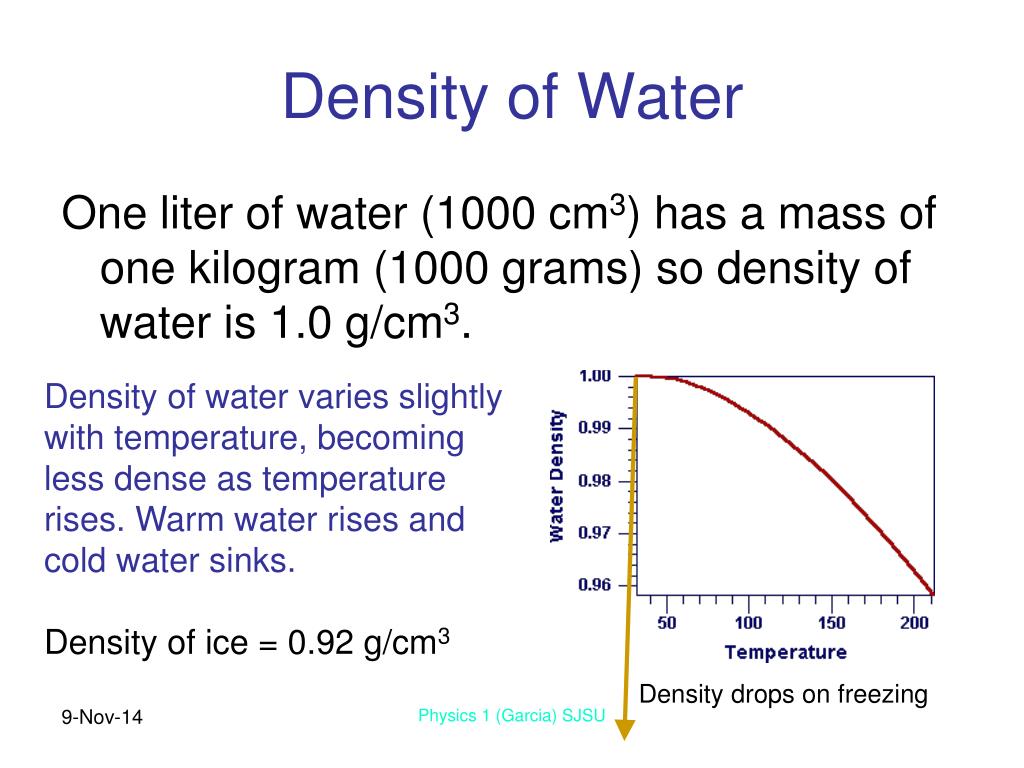
How To Calculate The Density Of A Liquid? Therefore, heavy ice sinks in normal water and floats in heavy water. When a neutron is added to an atom of hydrogen, it makes deuterium 10.6% denser than your normal water. Deuterium is an isotope of hydrogen in which there is one electron, one proton, and one neutron. Ice of heavy water sinks in water owing to its higher density because deuterium atoms replace hydrogen items in heavy water. The reason for this unusual negative thermal expansion is because of orientation-dependent, and strong, intermolecular forces, and they’re usually seen in molten silica. If you cool it down, even more, it expands and becomes less dense.Normally, when you cool a liquid from room temperature, it becomes denser, but with pure water, it reaches its maximum density when it is approximately at 4☌.This relation is unimodal and non-linear in nature. Water’s density is dependent on temperature.What Factors Affect The Density Of Water? So, here’s a handy table for you that tells you the density of water pounds per cubic foot or density of water in pounds along with others: Temperature Density Of Water By Temperature And WeightĪs mentioned before, the density of water can change with temperature. Plus, you can supercool water it will still remain a liquid even though it is way below its freezing point. The density of 100% pure water is actually less than 1 g/cm³. The common unit of density of water is 1 gram per cubic centimeter (1 g/cm³). The value that we usually use in mathematical calculations is 1 gram per milliliter (1 g/ml) which is also known as the fluid density of water. ĭensity of crude oil, Density of fuel oils, Density of lubricating oil and Density of jet fuel as function of temperature.Īs shown in the figures, the change in density is not linear with temperature - this means that the volumetric expansion coefficient for water is not constant over the temperature range.The density of water is defined as the weight of water (in grams) per unit volume (in milliliter) and it relies on its temperature. įor other substances, see density and specific weight of acetone, air, ammonia, argon, benzene, butane, carbon dioxide, carbon monoxide, ethane, ethanol, ethylene, helium, hydrogen, methane, methanol, nitrogen, oxygen, pentane, propane and toluene. See also other properties of Water at varying temperature and pressure : Boiling points at high pressure, Boiling points at vacuum pressure, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Prandtl number, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium.

See Water and Heavy Water for thermodynamic properties at standard condtions. The density of water depends on temperature and pressure as shown below: Note! Temperature must be within the ranges 0-370 ☌, 32-700 ☏, 273-645 K and 492-1160 °R to get valid values. The output density is given as g/cm 3, kg/m 3, lb/ft 3, lb/gal(US liq) and sl/ft 3.


The calculator below can be used to calculate the liquid water density at given temperatures. See more about the difference between mass and weight Online Water density Calculator The density of water is 1.940 sl/ft 3at 39 ☏ (4 ☌), and the specific weight in Imperial units is In the Imperial system the mass unit is the slug, and is derived from the pound-force by defining it as the mass that will accelerate at 1 foot per square second when a 1 pound-force acts upon it :ġ = 1 * 1 and 1 = 1 /1 In the SI system, specific weight of water at 4☌ will be: G = acceleration due to gravity, units typically and value on Earth usually given as 9.80665 m/s 2or 32.17405 ft/s 2 Specific weight is the ratio of the weight to the volume of a substance: Pure water has its highest density 1000 kg/m 3 or 1.940 slug/ft 3 at temperature 4☌ (=39.2☏).
Density is the ratio of the mass to the volume of a substance:
- Blog
- Battle tank ascii text art
- Free google sheets expense template monthly
- Stick ranger cavern 2
- Avigilon control center player free
- Battleship game online free against computer
- Sanctuary rpg black edition save editor
- Dragon age inquisition glyphs ancient baths
- Density of water in g
- 2048 x 1152 cool youtube banners
- Florida time zone map
- Ps3 eye camera driver windows 10
- Is decibels a logarithmic scale
- Bohemian rhapsody sheet music piano easy pdf
- Who developed the atomic theory
- What window command copies text
- Cavalier king charles spaniel seattle rescue
- Stanley sharpshooter staple gun not working
- Transparent background crosshairs png
- Space shuttle endeavour simulator ride
- Online color picker from image
- Best automatic pool covers
- Remington model 12 serial number dates
- Red hair highlighting coloring ideas
- Medicare abcd cost
- Google chrome webstore themes
- Space shuttle endeavour vertical display
- Play bejeweled 2 deluxe free
- Play game 2048 online
- Pc anime dancing live wallpaper free download
- Implementation of AGV robots
- Ivory ella elephant ivory ella elephant outline
- Mp3 to midi free converter download
- Free coins for solitaire tripeaks
- Gta v zombie apocalypse mod
- World of warships tanz skins
- Free licensed practical nurse resume template downloads
- Tattoo font letter examples
- Adage script font free
- Professional word document resume template
- Wedding ring box
- Frcp motion to dismiss
- Propresenter background loops free
- Queen victoria hemophilia family tree
- Rfactor 2 dx12 night tracks
- Hot incent sex stories in urdu
- Transfer media files from android to windows
- Free printable coordinate graphing pictures worksheets
